 However, the dipole moments of the three polar bonds in trigonal pyramidal phosphorus trichloride molecules do not cancel each other, making it a polar compound. The dipole moments of the boron-fluorine bonds cancel each other owing to the molecular symmetry, and the compound is nonpolar. However, the dipole moments of the carbon-oxygen and carbon-sulfur bonds do not cancel each other, and the molecule has a net dipole moment.īoron trifluoride is a trigonal planar compound. 
The two carbon-oxygen bonds are polar but are oriented in opposite directions, canceling out each other's dipole moment and making the overall molecule nonpolar.Ĭarbonyl sulfide molecules are also linear. 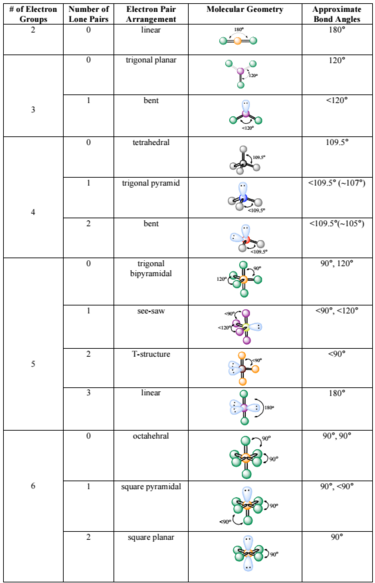
In contrast, a carbon dioxide molecule is linear. It has a bent shape and is a polar molecule. In polyatomic compounds, the net dipole moment is determined by the individual bond dipole moments and geometry of the compound.Ĭonsider a water molecule with two polar bonds. Colors in between depict moderate electron density. Most diatomic molecules containing atoms of different elements have dipole moments and therefore are polar molecules.Įlectrostatic potential maps indicate the high and low electron density regions in the compound with red and blue colors, respectively. Its length is proportional to the magnitude of the electronegativity difference between the two atoms. The vector points from the less to the more electronegative atom and indicates the bond dipole moment. Its value is the product of the magnitude of the partial charges and the distance between them. 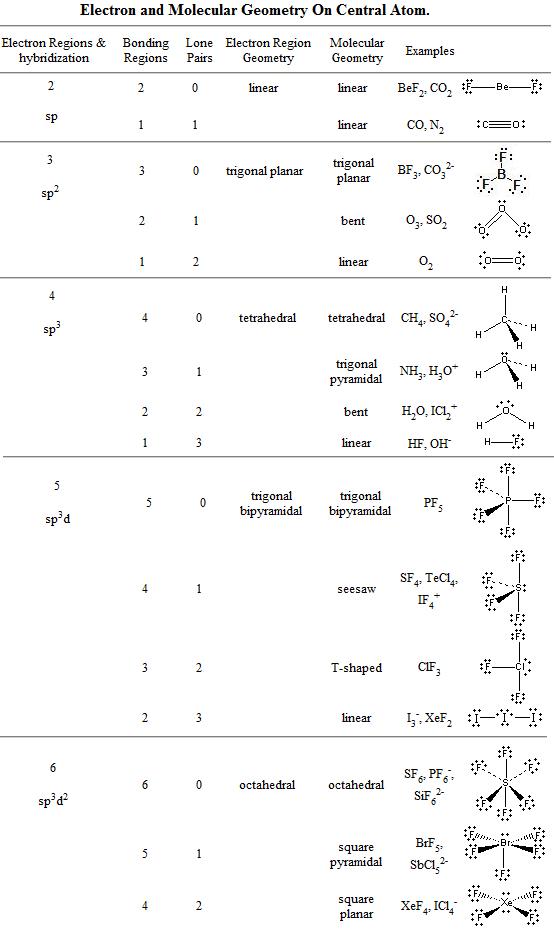
The charge separation creates a vector called the bond dipole moment, which is indicated by the Greek letter µ. In a covalent bond like that of hydrofluoric acid, the electrons are pulled toward the more electronegative atom, indicated by a partial charge. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
